Bone Marrow Microenvironment on a chip for culture of functional hematopoietic stem cells
Can organ on a chip platforms be used to culture and maintain functional hematopoietic stem cells and progenitor cells (HSPC)? This is a recent paper from Sharipol et al. from the University of Rochester, culturing important components of murine bone marrow microenvironment (BMME) in an organ on a chip. They used a commercial organ on a chip from Emulate Bio to co-culture osteoblasts, endothelial cells, mesenchymal stem cells, and HSPCs for up to 14 days. Afterwards, they performed competitive transplant assay to test the self-renewing ability of the HSPCs.
Results
The authors differentiated primary mouse bone marrow stromal cells (BMSC) under continuous perfusion of differentiation media. The monolayer showed mineralization with clear calcium nodules. They also showed that endothelial cells expressed tight junctions after 7 days of continuous flow. After 7 days of co-culture with the osteoblasts, the HSPC were maintained in the chip in greater population than in vivo. Furthermore, the content of the bone marrow environment in the chip was transplanted into mice. The engraftment produced similar levels of peripheral blood cells compared to control mice.
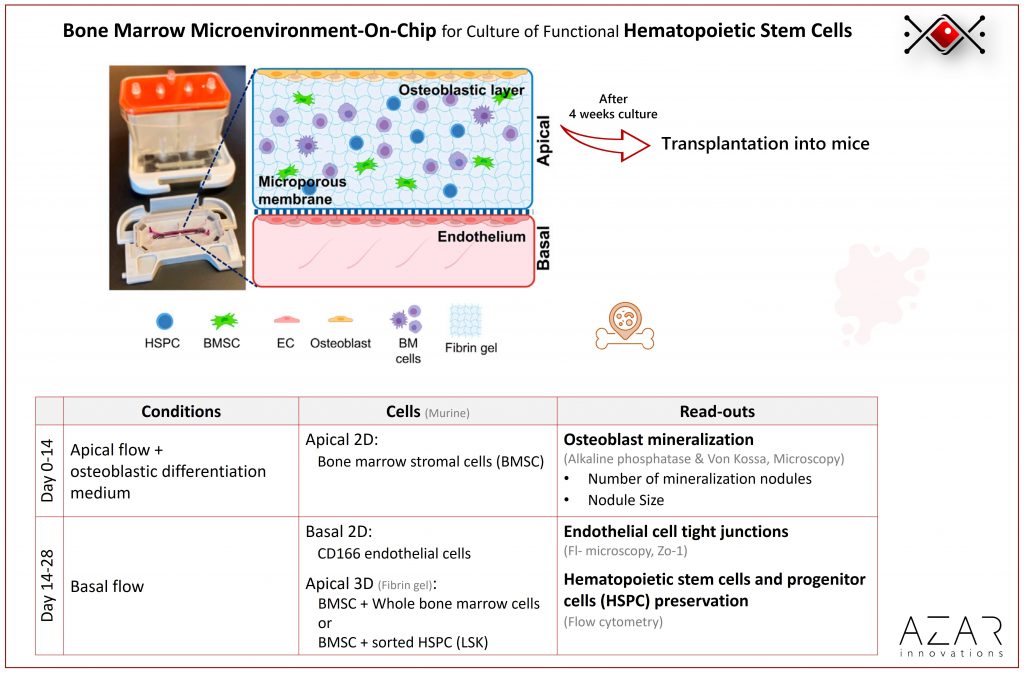
Method
Membrane chips with apical (top) and basal (bottom) channels were used. An osteoblastic layer was differentiated from primary mouse bone marrow stromal cells (BMSC) for 14 days under flow. Afterwards, bone marrow and stromal cells suspended in a fibrin hydrogel were introduced to the top channel. Simultaneously, a supportive endothelial layer was generated in the basal channel.
Fabrication: Commercially available organ on a chip from Emulate Bio
Sterilization: Ready to use from Emulate Bio
Cells incorporation: BMSC were isolated from mice and injected to top channel via the channel inlet. The chips were immediately flipped 180 degrees to result in a 2D mono-layer on the ceiling of the top channel (2 h incubation). Endothelial C166 cell line were injected to the chip and culture on the ceiling of the bottom channel with the same flipping method. The mixture of bone marrow cells and fibrin gel was added to the apical channel and incubated to polymerise.
Perfusion/refreshing method: Pressure-based pump by Emulate Bio (change medium in reservoirs every 2-3 days)
On-chip read-outs: End-point microscopy (fix and stain cells in the chip)
Off-chip read-outs: Flow cytometry (Dissociate and aspirate cells from the chip)
Strong points:
+ Automatic perfusion in both channels
+ Relatively long-term culture
+ Well-established commercial protocols
Improvement points:
– Although a proof of concept, human cells can better represent the physiology of the human body
– Not clear how the medium for the cells in the apical channel is refreshed
– Would have been interesting to study the role of endothelial cells in the functionality of the HSPCs
– A comparison of the mechanical properties of the fibrin gel to in vivo
Conclusions and outlook
This study’s bone marrow on a chip recapitulates the in vivo bone marrow microenvironment under controlled and dynamic conditions. This organ on a chip system can be used as a powerful tool to study bone marrow pathologies and test new therapeutics.
Do you have questions about the system in this paper or similar technologies? Or do you want to start with organ on a chip? Feel free to contact us!